Original article: Metal–Organic Frameworks for Cultural Heritage Preservation: The Case of Acetic Acid Removal
The ouroboros—an ancient symbol depicting a serpent eating its own tail (Figure 1)—is not only a powerful reminder of the cyclicity of life and death, it’s also a fitting representation of ‘vinegar syndrome.’ Many materials found in museums (wood, paper, film stock, and some plastics) produce acetic acid (the acidic component of vinegar) when they degrade. If not removed from the immediate environment of the collection, the acid can then attack those same materials and speed up their degradation. And so, the snake begins to eat its own tail. Acetic acid can cause paper to become brittle, certain metals and alloys to corrode, calcareous materials like stones and ceramics to degrade, and entire film collections to liquefy. Although the ouroboros illuminates the creation of life from death, conservation scientists care too deeply for cultural heritage objects to let them rapidly degrade of their own volition.
 Figure 1. Selection from ‘Putto with an ouroboros’ by Giovanni Cattini (attributed to), 1735-1800. Rijksmuseum, The Netherlands.
Figure 1. Selection from ‘Putto with an ouroboros’ by Giovanni Cattini (attributed to), 1735-1800. Rijksmuseum, The Netherlands.
For the sake of preserving our cultural heritage, it is critical that acetic acid be removed from the gallery environment as soon as it is produced. This is what Dedecker et al. have set out to do using molecular organic frameworks, or MOFs. MOFs—ultraporous materials with easily tailored structures allowing for tunable pore size and shape, surface area, and hydrophilicity/hydrophobicity—are known to capture a range of volatile organic compounds (commonly referred to as VOCs). The museum environment is a tightly controlled affair, with strict guidelines for temperature, relative humidity (RH), and VOC content. The 40% RH content required in most galleries means that traditional sorbents are overwhelmed by water rather than acetic acid, which is present in much smaller concentrations (ideally, a sorbent will capture acetic acid even when it is present in the low ppb range). By designing a technology to selectively capture acetic acid over water in the environment, Dedecker et al. provide a possible solution for slowing the degradation of materials susceptible to vinegar syndrome.
The researchers explored a series of MOFs with different chemical functionalities (selected because they promote binding with acetic acid) and hydrophobicities/hydrophilicities (Figure 2) and discovered that two MOF structures were capable of very efficiently removing acetic acid without becoming overwhelmed by the amount of water molecules in the air: UiO-66-2CF3 and MIL-140B. These MOFs lie in a hydrophilic ‘sweet spot’: hydrophilic enough to efficiently attract acetic acid, but not too hydrophilic to adsorb significant amounts of water. These two MOFs also have pores that are just the right size to capture and retain acetic acid in a very hospitable guest-host interaction.
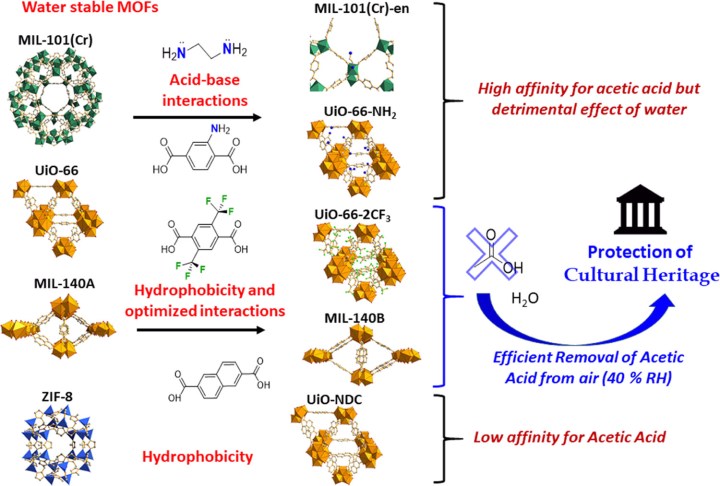 Figure 2. Four molecular organic framework structures, with and without chemical modifications, were explored for removing acetic acid from air. Color coding: Zirconium polyhedra (yellow), Chromium octahedra (green), and Zinc tetrahedra (blue); oxygen (red), nitrogen (blue), fluorine (green), and carbon (gray), with hydrogens omitted for simplicity. Reproduced with permission.
Figure 2. Four molecular organic framework structures, with and without chemical modifications, were explored for removing acetic acid from air. Color coding: Zirconium polyhedra (yellow), Chromium octahedra (green), and Zinc tetrahedra (blue); oxygen (red), nitrogen (blue), fluorine (green), and carbon (gray), with hydrogens omitted for simplicity. Reproduced with permission.
MOFs have three-dimensional scaffold-like structures: MIL-140B has a hydrophobic cage-like structure, while UiO-66-2CF3 is channel-like, with oil-loving -CF3 groups along its cavities. After experimentally selecting the best MOF for the job, the group ran Monte Carlo computational simulations to find out how the acetic acid and water molecules adsorb into their pores (Figure 3). Their model shows that the -CF3 groups create a high affinity for acetic acid in the MOF.
 Figure 3. Monte Carlo simulations of acetic acid and water in the pores of the two molecular organic frameworks that captured acetic acid most successfully. Adapted from the original article.
Figure 3. Monte Carlo simulations of acetic acid and water in the pores of the two molecular organic frameworks that captured acetic acid most successfully. Adapted from the original article.
At the end of their article, the authors even hint at the holy grail of selective sorbents for museums: their MOFs might efficiently and selectively remove other harmful VOCs like acetaldehyde, AND be re-usable when recovered through thermal regeneration. Not only does this work highlight how MOFs can be modified to selectively capture VOCs, it details a promising innovation for the preservation of cultural heritage during display and storage. It will be interesting to see how these materials perform (in terms of both stability and acetic acid uptake) over long periods of time and, ideally, in a real-world, museum setting.
All figures reproduced/adapted with permission from ACS Appl. Mater. Interfaces 2018, 10, 13886−13894. Copyright 2018 American Chemical Society.

Fascinating article on fascinating complexes!
LikeLike