Original article: Facile Synthesis of Three Kobolds: Introducing Students to the Structure of Pigments and Their Characterization
How often do we discover a brand new color? Well, not very often these days, but that hasn’t always been the case! Throughout history, people have always been fascinated by new, flashy colors. Beautiful, exotic minerals were discovered in the Old World during explorations and then ground up to make natural pigments. Later, the Industrial Revolution saw the development of complex syntheses for vibrant Impressionist pigments. Even now, Crayola continues to introduce new eco-friendly crayons to its collection each year.
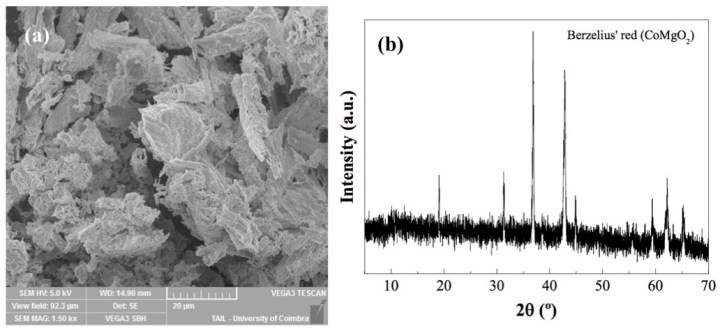
Cobalt blue (CoAl2O4) is one such popular, synthetic pigment developed to meet the growing demand for new vibrant colors during the Industrial Revolution. It was discovered by Louis Jacques Thénard in 1802, and for that reason, it is also referred to as Thénard’s blue. Other cobalt oxide synthetic pigments include Rinman’s green (CoZnO2), which is a semitransparent, bright green, and Berzelius’ red (CoMgO2), which is pale red to pink in color. Small changes in the atoms or in their arrangement can cause drastic changes in a compound’s color, as shown in Figure 1. Co(II)-based pigments originated in mines referred to as “kobolds,” a word that originates from the word Bohemian miners used for ghosts; they believed that the mines were haunted by spirits, which caused illnesses and troubles. These cobalt pigments were used throughout the 19th century in famous artworks with Thénard’s blue being the most common of the three. Thénard’s blue was used by Claude Monet, Pierre-Auguste Renoir, and Vincent van Gogh, including Starry Night Over the Rhone (1888)!

Traditionally, these compounds are formed by firing in a furnace (or pyrogenesis). This ceramic method involves milling an oxidized cobalt salt with alumina, zinc oxide, or magnesium oxide before firing. In this paper, the authors suggest a modified version of synthesis that can be done easily in a laboratory: after soaking filter paper in a cobalt nitrate (Co(NO3)2) solution, the filter paper is dried and a solution of Al3+, Zn2+, or Mg2+ salts is added, depending on the desired color. The filter paper will change color, but after igniting the paper, an ash-like, more strongly-colored pigment is produced.
In the activity, the synthesized pigments are then characterized using two different techniques: X-ray powder diffraction (XRPD) and scanning electron microscopy energy-dispersive X-ray spectroscopy (SEM-EDS). XRPD determines the structural properties of crystalline materials based on how they diffract incident X-rays (Figure 2b). SEM uses an electron beam to generate high-resolution images of samples; this method is useful for observing the texture of pigments (Figure 2a). SEM can be coupled with energy dispersive X-ray spectroscopy (EDS), in which the incident electron beam displaces electrons in the sample from their usual energy level. The resulting electron transitions cause the generation of characteristic X-rays, which semi-quantitatively tell us which elements are in our sample.
This activity was designed for chemistry and science undergraduates or conservation and art students, but the authors stress the adaptability of this experiment for a variety of audiences. Like many activities involving conservation materials, these may be easily modified to engage a wide variety of people from different disciplines, backgrounds, and age groups! The materials are easily prepared in a lab or at an outreach event. Instead of characterizing the pigments, they can be mixed with binders (such as egg yolks or linseed oil), and then participants can make their own artwork. It’s also possible to skip the chemistry lesson altogether and focus on the folklore or the historical applications of the pigments. Conversely, the materials’ characterization aspect could be emphasized, and other techniques, such as Raman or infrared spectroscopy, could be introduced, based on availability.
Martín-Ramos, et al. have used Co(II)-based pigments in this interdisciplinary combination of art and chemistry in an introduction to the world of conservation science from synthesis to analysis. They write that this activity was well-received by the students, and it is excellent proof that there are constantly new opportunities to highlight conservation science and artists’ materials in education!
All figures reproduced/adapted with permission from J. of Chem. Edu. 2018 95 (8), 1340-1344. Copyright 2018 American Chemical Society.

One thought on “Cobalt pigments as teaching tools”